SoftComply
Risk Manager Plus on Cloud User Guide
_toc QUICK START GUIDE Here is a short video tutorial walking you through the steps listed…
SoftComply
Risk Manager User Guide
_toc QUICK GUIDE TO THE RISK MANAGER Success! You have just created your first risk management…
SoftComply
Risk Manager Plus User Guide
_toc QUICK START GUIDE Success! You have just created a new risk management project and you…

Software Risk Analysis in Medical Devices
In the Medical Device industry software components, whether standalone or as part of a physical device,…

Risk Management of Off-The-Shelf (OTS) Software
Your software is likely to contain a number of components, items, packages, libraries, etc. not developed…
Hazard Analysis and FMEA: the Most Common Approaches to Risk Management for Medical Devices
What is the difference between Hazard Analysis and FMEA. Which approach should you use. Comparison and examples.

BOOST YOUR PRODUCTIVITY & TRANSFORM YOUR RISK REGISTER IN JIRA
Why Jira for your Risk Register? Risk Management is an essential governance practice for enterprise, product,…

3 Best Jira Cloud Risk Management Plugins Compared [2024]
Comparison of risk apps on Jira Cloud

In Search of a Compliance Solution that Lets You Sleep at Night
How to find the best regulatory compliance solution for medtech on Atlassian
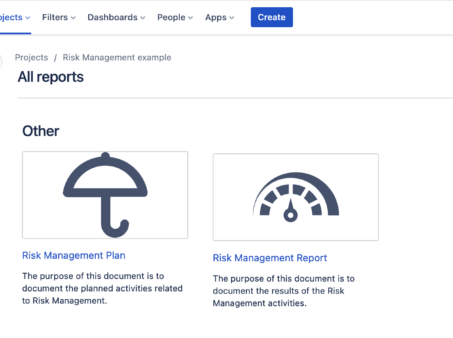
Risk Management Guide for a Digital Health or a Medical Device Company on Jira Cloud
Why Risk Management In the fast-paced world of medical device development, safety and quality are of…

How to Build a Risk Analysis in Jira
How to build a risk analysis in Jira with the SoftComply Risk Manager app
SoftComply
Why Decompose the Probability of Harm in two – P1 and P2
P1 and P2 No we are not talking about the early Playstation versions… ISO 14971 (2019…