
NIS2 and DORA Compliance and Protecting your Atlassian Cloud Data
This is a guest-article from Atlassian Marketplace Partner, Revyz In late 2022, the European Parliament introduced…

Using Jira for Risk Management: 8 Best Practices For 2024
When it comes to managing risks in project management, using Jira for risk management can significantly…
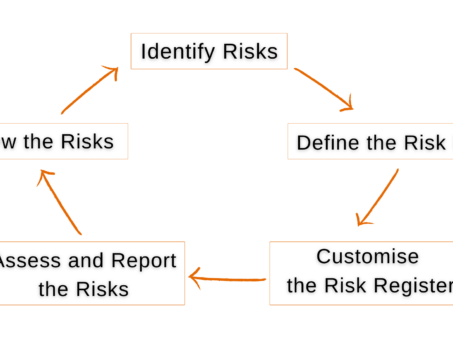
5 Steps To Tracking & Mitigating Project Risks in Jira
Project risk management is crucial for project success, especially important to keep critical risks under check…

Supporting Regulated Industries on Atlassian
On May 2, 2024 SoftComply hosted the 2nd edition of Regulated Industries workshop during Atlassian Team…

What are the FDA 21 CFR 11 Compliant Electronic Records?
Title 21 of the Code of Federal Regulations, Part 11, also known as 21 CFR 11,…

What is an FDA 21 CFR 11 Compliant Electronic Signature?
Title 21 of the Code of Federal Regulations, Part 11, also known as 21 CFR 11,…

Software Risk Analysis in Medical Devices
In the Medical Device industry software components, whether standalone or as part of a physical device,…

Risk Management of Off-The-Shelf (OTS) Software
Your software is likely to contain a number of components, items, packages, libraries, etc. not developed…
Hazard Analysis and FMEA: the Most Common Approaches to Risk Management for Medical Devices
What is the difference between Hazard Analysis and FMEA. Which approach should you use. Comparison and examples.

4 Key Reasons Why you should move your Risk Management to Jira
Risk management in the regulated industries is often viewed as a necessary evil, but what if…

4 Steps to ISO 27001 Compliance – Start with Risk Management in Jira
Being an ISO 27001-compliant company means that you protect your information assets and client data against…

3 Simple Steps to Prioritising your Critical Risks
“It’s a dangerous business, Frodo, going out your door. You step onto the road, and if…